Therapeutic vs maintenance dosing
Therapeutic dosing: Higher doses, often more frequently throughout the day
Maintenance: Lower doses, often less frequently throughout the day
Immune-Specific Dosing
The concept of therapeutic and maintenance dosing is based on the theory of ‘trained immunity’ (1,2). This theory suggests that ongoing, high doses of ß-glucan-rich mushrooms are not, in fact, necessary to potentiate immune modulation and protection from infections. The innate immune system, or the “first responders,” has the ability to create non-specific memory responses to a variety of subsequent infections after a primary infection or vaccination. For example, a person may become infected with a specific pathogen (virus, bacteria or fungi) and the innate immune system will mount a response to that specific pathogen. Additionally, innate memory cells will remain that can help protect against that same infection in the future, as well as other unrelated infections, i.e. a non-specific memory response. These changes in immune memory have been observed for as long as one year following initial administration of ß-glucans (3). These innate memory cells have a relatively short lifespan, and so it seems unlikely that trained immunity would last as long as it does. However, researchers have discovered that ß-glucans not only induce trained immunity in mature innate immune cells, but they also alter the cells hematopoietic progenitors (cells that will eventually differentiate into innate immune cells) within the bone marrow leading to the prolonged presence of trained monocytes and macrophages in circulation (4). The ß-glucans are essentially playing a role in your immune cells’ epigenetic purpose.
Initial higher dosing of ß-glucan-rich mushrooms is like an immune training program. Similar to building muscle, the initial training takes a little longer, and you have to train more often. Maintaining muscle is much easier than building muscle, and sometimes once a week training is all that is needed for muscle maintenance. Similarly, taking mushrooms one to two times per day, and maybe even a few times per week, is all that is needed for immune maintenance.
Initial dosing during the therapeutic phase for immune modulating effects should be done repeatedly, ideally three times per day. One study comparing the duration of the effect of oral dosing versus intravenous dosing of ß-glucans found that the repeated oral dosing “clearly resulted in stronger and longer action stimulation (5).” Intravenous dosing of ß-glucans is mostly available in clinical trials outside of the United States and is not yet common practice in the U.S. Therefore, repeated oral dosing two to three times per day is most likely to optimize initial immune benefits.
Neurocognitive-specific Dosing
When dosing mushrooms like lion’s mane, tremella and oyster mushroom for neurocognitive benefits, the idea of immune training or cellular training does not apply. In the majority of studies exploring the neurocognitive benefits of lion’s mane, researchers have found that many benefits relating to memory and mood were no longer present after discontinuation of supplementation. It is important to note, however, that certain patterns related to mood may be a result of “negative neuroplasticity.” Negative neuroplasticity is the idea that getting stuck in a particular maladaptive pattern, like insomnia, can create neural patterns that are then primed for insomnia. Changing certain behaviors or taking certain medications and/or supplements can help to rewire negative patterns. Mushrooms with neurological actions, such as BDNF-mediated neurogenesis, may help to rewire negative plasticity, and therefore may not be needed on a daily basis as previously thought. While this may be true for mood-related disorders, it is unlikely to be the case for memory and cognitive enhancement. Either way, once daily dosing seems adequate for neurocognitive effects.
Absorption
Mushrooms are composed of many different constituents. These constituents have different physiological activities that are fully dependent on whether they are absorbed, or not absorbed, into the bloodstream from the intestines.
Immune considerations
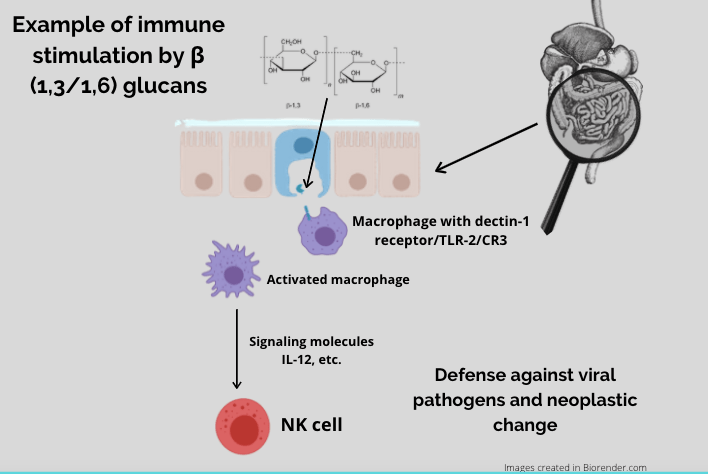
The immune-modulating benefits of mushrooms are dependent on a healthy environment within the small intestine, where there are groups of immune cells within the gut-associated lymphoid tissue (GALT). The GALT contains pockets of immune cells called Peyer’s patches which contain M cells, and M cells present ß-glucans to innate immune cells like macrophages and dendritic cells via specific cell-surface receptors, most notably dectin-1, but also TLR2 and CR3. Mushroom ß-glucans bind to dectin-1 and stimulate an immune response throughout the bloodstream, lymphatic system, spleen, bone marrow, and lymph nodes.
It is important to note, however, that not all ß-glucans are absorbed into the bloodstream, and that some make their way to the large intestine. Mushroom ß-glucans are prebiotics, acting as food for healthy gut microbes and improving the intestinal microenvironment (6). A healthy intestinal microenvironment is fundamental to human health and research has shown that microbial fermentation of fungal ß-glucans can increase important post-biotic short chain fatty acids like butyrate (7). Improving the intestinal microenvironment does not result in trained immunity, like small intestinal absorption of ß-glucans does, so for prebiotic benefits, daily dosing seems best.
Additional Considerations
Food. If there is a lot of food in the small intestine, there will be less surface area for the ß-glucans to come into contact with M cells, and the immune response will likely be less significant than it would be on an empty stomach.
Alcohol. Chronic alcohol consumption has been shown to alter the innate immune system at the level of the Peyer’s patch (8). Frequent alcohol intake may lessen the immune benefits of mushroom ß glucans.
Tannins/polyphenols. Blending and emulsifying ß-glucans with tannin-rich substances like coffee, tea and chocolate create a tannin-polysaccharide complex. However, the bonds between polysaccharides and polyphenols are hydrogen bonds and are easily impacted by temperature and pH (acidity). Bonds can be broken at high temperatures between 68°F-104°F, so if blending mushroom powders with tannin-rich foods, blend at higher temperatures rather than cold. If polysaccharides are contained in these complexes when they reach the small intestine, they will not be available to bind immune receptors in the GALT and their immune-modulating properties will likely be nullified (9).
Work Cited
- Moerings, B. G. J., de Graaff, P., Furber, M., Witkamp, R. F., Debets, R., Mes, J. J., van Bergenhenegouwen, J., & Govers, C. (2021). Continuous Exposure to Non-Soluble β-Glucans Induces Trained Immunity in M-CSF-Differentiated Macrophages. Frontiers in Immunology, 12. https://doi.org/10.3389/fimmu.2021.672796
- Garcia-Valtanen P, Guzman-Genuino RM, Williams DL, Hayball JD, Diener KR. Evaluation of trained immunity by β-1, 3 (d)-glucan on murine monocytes in vitro and duration of response in vivo. Immunol Cell Biol. 2017 Aug;95(7):601-610. doi: 10.1038/icb.2017.13. Epub 2017 Feb 23. PMID: 28228641; PMCID: PMC5550561.
- Moorlag SJCFM, Khan N, Novakovic B, Kaufmann E, Jansen T, van Crevel R, Divangahi M, Netea MG. β-Glucan Induces Protective Trained Immunity against Mycobacterium tuberculosis Infection: A Key Role for IL-1. Cell Rep. 2020 May 19;31(7):107634. doi: 10.1016/j.celrep.2020.107634. PMID: 32433977; PMCID: PMC7242907.
- Mitroulis I., Ruppova K., Wang B., Chen L.S., Grzybek M., Grinenko T., Eugster A., Troullinaki M., Palladini A., Kourtzelis I. Modulation of Myelopoiesis Progenitors Is an Integral Component of Trained Immunity. Cell. 2018;172:147–161.e12.
- Vetvicka, Vaclav & Vetvickova, Jana & Ms,. (2008). A Comparison of Injected and Orally Administered β-glucans. 11.
- Nowacka-Jechalke N, Juda M, Malm A. The preliminary study of prebiotic potential of Polish wild mushroom polysaccharides: the stimulation effect on Lactobacillus strains growth. Eur J Nutr. 2018 Jun;57(4):1511-1521. doi: 10.1007/s00394-017-1436-9. Epub 2017 Mar 28. PMID: 28353071; PMCID: PMC5959981.
- Mitsou EK, Saxami G, Stamoulou E, Kerezoudi E, Terzi E, Koutrotsios G, Bekiaris G, Zervakis GI, Mountzouris KC, Pletsa V, Kyriacou A. Effects of Rich in Β-Glucans Edible Mushrooms on Aging Gut Microbiota Characteristics: An In Vitro Study. Molecules. 2020 Jun 18;25(12):2806. doi: 10.3390/molecules25122806. PMID: 32570735; PMCID: PMC7355846.
- Lopez MC, Watzl B, Colombo LL, Watson RR. Alterations in mouse Peyer’s patch lymphocyte phenotype after ethanol consumption. Alcohol. 1997;14(2):107-110. doi:10.1016/s0741-8329(96)00104-8
- Li R, Zeng Z, Fu G, Wan Y, Liu C, McClements DJ. Formation and characterization of tannic acid/beta-glucan complexes: Influence of pH, ionic strength, and temperature. Food Res Int. 2019;120:748-755. doi:10.1016/j.foodres.2018.11.034
citations?