Fungi immune-modulation infographic

Fungi polysaccharides infographic

Oxalates and Fungi
Cause for concern?
The most common manifestation of kidney stones is without a doubt, calcium oxalate stones – 80% of stones, to be exact. For this reason, people who are prone to stones tend to avoid high oxalate-containing foods. The most popular oxalate-rich delicacies: spinach, beets greens, chard, chocolate, tea, nuts, grains, and yes, some mushrooms. It is important to keep in mind that not all oxalate-rich foods will lead to kidney stones – chemistry is much more complex than that.
There was a recent report1 about an older Japanese woman with liver cancer who consumed 4-5 tsp of Chaga mushroom per day for 6 months, and happened to get nephrotoxicity. The postulated culprit: Chaga mushroom. Unfortunately, in this study, there was neither mention of the source of the Chaga, if this was the canker or mycelium, nor if the Chaga powder was an extract or not. This case report concluded that it must have been the high oxalate content in the Chaga mushroom that induced this nephrotoxicity. Is this a true cause or simply a correlation? Should consumers be concerned about the oxalate content in in all Chaga products?
My hope is that this post will be educational, and readers will walk away with some answers to these questions.
This report got me thinking, about Chaga and oxalates and wondering, could this really be? I mean, they do look so similar. Could this be an example of the doctrine of signatures or is it possible that if you crush any golden substance into small enough pieces it may resemble calcium oxalate fragments? (No offense to the doctrine of signatures)
Some education about oxalates
First, not all oxalates are created equal. There are soluble and insoluble oxalates. Insoluble oxalates are already bound to minerals, for example: calcium oxalate, magnesium oxalate and iron oxalate. The insoluble oxalates in foods pass right into the GI and come out in feces. They will not be absorbed in the blood stream, and so they are not a concern for causing hyperoxaluria (high urine oxalates). Examples of soluble oxalates are potassium and sodium oxalate. Unlike soluble oxalates, these acids release free oxalate anions which can pass into the blood stream. Free oxalate can then bind with free calcium and make calcium oxalate crystals;
This distinction is important to keep in mind when considering high oxalate foods.
Mushrooms and Oxalates
White button mushroom and Shiitake mushroom are both very high in oxalates, but 99% of them are insoluble! So, don’t fret about these mushrooms – the oxalate will all come out in your shiit(ake).2

Oyster mushrooms, on the other hand, are moderately high in oxalates, and 90% of them are soluble. They contain a comparable amount to chocolate, almonds, and grains.2 So if you can eat more oyster mushrooms than you can chocolate, you are my hero and you may want to eat them with some calcium containing foods – more about this later.

Lucky for kidney stone formers, they can continue to consume large quantities of Lion’s Mane (Hericium erinaceus), Cauliflower mushroom (Sparassis spp) and Reishi (Ganoderma spp). There are no soluble oxalates found in any of these mushrooms.2
Remember, for an organism to take the time and energy to make a molecule, the molecule must serve some purpose for that organism; oxalates are not made to cause kidney stones in humans. I hope to find more on this subject in relation to mushrooms, but for now, I found one study. According to this study, calcium oxalate crystals form in response to toxic metal stress.3 So perhaps mushrooms growing in environments where there are more toxic metals will in turn have a higher calcium oxalate load than those same species growing elsewhere.
Oxalates and Chaga
Oxalate samples were assessed in Chaga samples from Russia, Finland and Thailand. A hot water extract was made of the Chaga canker (the growth coming out of the tree, traditionally used as medicine) and oxalate content was assessed. Researchers found that for 1 gram of the Russian Chaga extract there was 97.6mg of soluble oxalic acid and 24mg of insoluble oxalic acid. The Finnish Chaga extract contained 55.62mg/g of soluble oxalates and 9.5mg/g of insoluble oxalates. Japan Food Research Laboratories measured oxalate content of the mushroom powder and concentrations of 11.2g total oxalate in 100g of powder. Another test from this laboratory on a separate Chaga sample was 2.8g/100g.1
Some perspective: These amounts are higher than most edible mushrooms, similar to the amount in almonds, peanuts, cereal grains and chocolate, and much lower than what is found in high oxalate foods like spinach, rhubarb and beet greens.4
Cooking and Oxalates
Oxalate load was compared in a 10g sample of dried mushrooms cooked versus not cooked. This study found that cooking slightly decreases oxalates, but also seems to increase them in certain cases. Interestingly, levels of soluble oxalate from forest harvested mushrooms was generally lower than cultivated mushrooms.2
Concerned about your mushroom intake and risk of kidney stones?
Here is some food for thought:
Calcium intake and oxalate absorption
People with low calcium diets can benefit from consuming calcium-containing foods along with oxalate-containing foods to decrease oxalate absorption. One study found that exogenous (dietary) calcium leads to a linear reduction of oxalate absorption within the range of daily intake of 200-1200mg of calcium. This case has been documented in numerous studies5,6,7 and calcium supplements have been shown to lower the oxalate absorption. One study assessed this theory using 1000mg calcium supplement given simultaneously with soluble oxalates. They concluded that this technique will reduce oxalate absorption by 10%, however this is mostly effective if the person already has a low calcium diet. When calcium is supplemented to an already high calcium diet, there is only a 1% decrease in oxalate absorption.

Perhaps this is an excellent reason to add milk to tea and coffee and of course, to your favorite mushroom drink. To give some perspective, 1 cup of dairy milk has about 305mg of calcium and 1 cup of fortified almond milk has about 300mg of calcium.
The microbiome and oxalate absorption
Within the microbiome resides a bacteria known as Oxalobacter formigenes.8 This superlative gut bacteria degrades oxalates and helps to prevent hyperoxaluria and kidney stones. So, for those concerned about the oxalate content in the mushrooms discussed, perhaps this little bacteria is your ally. Studies looking at oral intake of Oxalobacter have shown less urinary oxalate excretion following administration of an oxalate heavy food load compared to when there was no Oxalobacter administered. Unfortunately, this bacteria is susceptible to antibiotic use. Specifically, clarithromycin and doxycycline have been shown to annihilate this little probiotic, leaving the human left behind to be more susceptible to kidney stones.

Representation of Oxalobacter formigenes activity. Utilizes oxalate as a source of energy through the oxalate-formate antiporter.9
This is all very interesting and something most people do not need to worry about. Though I would say, if you are an oxalate kidney stone former: don’t eat a ton of oyster mushrooms or drink multiple cups of Chaga tea per day. Do make sure to drink plenty of water and eat a diet with adequate amounts of calcium, and hope that antibiotic use hasn’t eradicated all of your Oxalobacter.
Work Cited
- Kikuchi Y, Seta K, Ogawa Y, et al. Chaga mushroom-induced oxalate nephropathy. Clin Nephrol. 2014;81(6):440-444. doi:10.5414/CN107655
- Savage GP, Nilzen V, Österberg K, Vanhanen L. Soluble and insoluble oxalate content of mushrooms. Int J Food Sci Nutr. 2002;53(4):293-296.doi:10.1080/09637480120057000
- Jarosz-Wilkolazka A, Gadd GM. Oxalate production by wood-rotting fungi growing in toxic metal-amended medium. Chemosphere. 2003;52(3):541-547. doi:10.1016/S0045-6535(03)00235-2
- Glamočlija J, Ćirić A, Nikolić M, et al. Chemical characterization and biological activity of Chaga (Inonotus obliquus), a medicinal “mushroom.” J Ethnopharmacol.2015;162:323-332. doi:10.1016/j.jep.2014.12.069
- Von Unruh GE, Voss S, Sauerbruch T, Hesse A. Dependence of oxalate absorption on the daily calcium intake. J Am Soc Nephrol. 2004;15(6):1567-1573.doi:10.1097/01.ASN.0000127864.26968.7F
- Bong WC, Vanhanen LP, Savage GP. Addition of calcium compounds to reduce soluble oxalate in a high oxalate food system. Food Chem. 2017;221:54-57. doi:10.1016/j.foodchem.2016.10.031
- Brogren M, Savage GP. Bioavailability of soluble oxalate from spinach eaten with and without milk products. Asia Pac J Clin Nutr. 2003;12(2):219-224.
- Duncan SH, Richardson AJ, Kaul P, Holmes RP, Allison MJ, Stewart CS. Oxalobacter formigenes and its potential role in human health. Appl Environ Microbiol.2002;68(8):3841-3847. doi:10.1128/AEM.68.8.3841-3847.2002
- Corica, Domenico & Romano, Claudio. (2015). Renal Involvement in Inflammatory Bowel Diseases. Journal of Crohn’s & colitis. 10. 10.1093/ecco-jcc/jjv138.
- Oxalates in Chaga – A Potential Health Threat By Michael W. Beug, Chair NAMA Toxicology Committee
- Mitchell T, Kumar P, Reddy T, et al. Dietary oxalate and kidney stone formation. Am J Physiol Renal Physiol. 2019;316(3):F409–F413. doi:10.1152/ajprenal.00373.2018
Lion’s Mane: a Psychosomatic Psychobiotic
I know you immediately think of the brain, but follow the vagus nerve from brain to gut and let’s just stay there for a while…

This mushroom is incredibly popular right now. Very hip. Very trendy.
Also of grand popularity are afflictions of the stomach, intestines and the colon. Many people have a visceral response to this overstimulating existence of modernity, and the most common viscera affected – the gut. Not only is the entire weight of the world living there, but the once-prolific community of microorganisms that once inhabited this space, and that would normally support the gut with the physiological burden that comes with this weight, has been significantly depleted. The reasons for this lack of microbial support are many, though we can just call it an unhealthy obsession with purity. The problem here is that this physiological burden that comes with the stress of the world, more often than not, becomes a pathology. Lion’s Mane extracts infiltrate the gut and the associated lymphoid tissue, regulating inflammation, decreasing oxidative damage, feeding the residing microbiota and modulating the immune system – all desired actions for healing, and hopefully avoiding, this potential pathology. Before being known as a nootropic medicinal, this mushroom was used in Traditional Chinese Medicine as a digestive tonic and to support gastrointestinal health. I have gone through the most current research to support the use of Lion’s Mane for gut health, and so overall human health.
Warning – most all of the research has been done on rats or mice. This is just the reality of most in vivo research and that is all we get until human clinical studies, which are incredibly difficult to find funding for – especially with natural products that don’t have potential to be synthesized into pharmaceuticals – can be done. So if you need to blame someone, blame capitalism and big pharma.
Rodent studies exploring Hericium Extracts and inflammatory bowel disease (IBD)
There have been many studies exploring variations of Lion’s Mane extracts and the effects of these extracts on rodents with chemically induced inflammatory bowel disease (I know it is fucked up). In one study1, mice were exposed to 2% dextran sulfate sodium (DSS) for 7 days to induce acute intestinal inflammation. These mice were then administered an ethanol extract of Hericium at a dose of 250mg/kg/day (This is a very big dose). Results showed that the treated mice had significant improvement in body weight, colon length and overall decreased intestinal bleeding compared with control mice. After administration of the extract, there was suppressed production of inflammatory mediators (TNF-alpha, IL-1â and IL-6) in colon tissue, as well as a decrease in oxidative damage potentiators (nitric oxide production, malondialdehyde, and superoxide dismutase). Overall, Lion’s Mane decreased inflammation and oxidative stress, leading to a decrease in mucosal damage and protection of mucosal epithelium.
Another in vivo study2 induced colitis in mice using this same DSS solution, and found similar results with Hericium polysaccharides. In this study, researchers tested markers of inflammation and oxidative stress before and after administration of the polysaccharides. Similar to the previously described study, there was a marked improvement in clinical symptoms postulated to be due to the decrease in nitric oxide, malondialdehyde, superoxide dismutase and myeloperoxidase. There was also a significant reduction in inflammatory cytokines TNF alpha, IL-1ß, and IL-6. Other notable markers decreased after polysaccharide administration include COX-2 and iNOS. COX-2 is an important enzyme in the prostaglandin synthesis pathway and the target of NSAIDs (like aspirin). Reduction or inhibition of this enzyme leads to decreased pain and inflammation. Intriguingly, this extract also blocked phosphorylation of NF-kappaB, p56 and also reversed DSS- induced gut dysbiosis and maintained intestinal barrier integrity. NF-kappaB is the major transcription factor for inflammatory cytokine production and if it is not phosphorylated it will not be active, and so less overall inflammation. (I realize I am really driving home the idea that we want to reduce inflammation, and inflammation is NOT always bad, but in a condition like inflammatory bowel disease, where the inflammation is causing significant damage to the mucosa and destroying a person’s quality of life, inflammation is not desirable).
Another method of inducing colitis in rats is with a trinitrobenzene sulfonic acid (TNBS) enema. To further explore the therapeutic potential of various Hericium extracts on inflammatory bowel disease, another study3 had to be done, using rats with this TNBS induced colitis. After the TNBS enema, the rats exhibited symptoms of reduced activity, lethargy, weight loss, ruffled fur, and bloody stool. After 14 days of Lion’s Mane extract treatments, there was significant improvement in all symptoms. Serum levels of inflammation were also monitored during this time, and decreased significantly after treatment. The proportion of FOXP3 and IL-10 positive cells significantly increased, especially in the rats receiving the alcoholic extracts compared with the model group. FOXP3 is the transcription factor for T-regulatory cells, and T regulatory cells secrete IL-10, so increased levels indicate improved immune regulation.
Prebiotic activity: Another study4 using an IBD rat model explored immunomodulatory activity of the water soluble fungal protein, HEP3, extracted from the mushroom fruiting body. The mechanism proposed: regulation of the gut microbiota. The study explained that HEP3 is a protein, and its digestion and absorption is reliant on proteases and peptidases extracted from bacteria. The nitrogenous degradation products of protein digestion also serve as important nutrient and energy sources for some anaerobic organisms. Knowing this, HEP3 can significantly influence the diversity, structures, and metabolism of organisms and microorganisms. Evidence suggests that the diversity of gut microbiota is reduced in IBD patients, so finding treatments that can target both inflammation and increase biodiversity is ideal. In this study, after HEP3 administration, Bifidobacterium abundance increased significantly and the colon tissue damage, inflammation, other prebiotics and diversity and structures improved significantly. Bifidobacterium is a beneficial genus of bacteria, associated with enhanced gut health and overall human health. This study concluded that HEP3 improves the immune system via regulation of the structure and metabolism of gut microbiota. The researchers postulate that through this prebiotic role, HEP3 activates the proliferation and differentiation of T cells and stimulates antigen presenting cells.
This prebiotic activity was also found in a hot water extract of the fruiting body.5 The preparation method was to simmer dried and ground mushroom in water (1:10, w:v) for 8 hours followed by pouring ETOH over the extract and leaving overnight. The alcohol was used to precipitate the polysaccharides which were then collected and used as the treatment in question. This method of precipitating polysaccharides out with alcohol is typically how polysaccharides are extracted in a laboratory setting, and so when people say that alcohol will destroy the polysaccharides in your mushroom extract, that is simply not true. This study found that Lactobacillus plantarum was the probiotic most affected by the Hericium polysaccharides. At first, after administration of the polysaccharides, the bacterial population rapidly increased after 6h. Also, the molecular weight of the polysaccharide decreased due to the loss of glycosidic bonds from gastrointestinal digestion, leading researchers to suppose that digestion is vital for the bioactivity of these polysaccharides in the GI system.
Immune modulation: Not only are these polysaccharides food for our gut bacteria, but they are also the primary constituent involved with immune modulation. There are many receptors in the phagocytes associated with mucosal immunity and the most often discussed regarding mushroom 𝛃-glucans is Dectin-1. Interestingly, one study6 found evidence that that the major pattern recognition receptors for Hericium polysaccharides are TLR2 and mannose receptor rather than Dectin-1. (fangfang wu 2017) In an in vivo mouse study assessing Hericium polysaccharides and their effect on cell mediated immunity (T-cells and phagocytes), humoral immunity (B-cells, complement and antimicrobial peptides), phagocytic capacity of peritoneal cavity phagocytes, and NK cell activity, results showed that Hericium polysaccharides improve immune function by functional enhancement of all of the above.

Neuroimmunity: mucosal immunity and vagus nerve regulation
The vagus nerve, the 10th cranial nerve and the queen of the parasympathetic response. This is the rest and digest, the chill and feel, the relax and pass response. The Vagus nerve also innervates the majority of our digestive organs, hence the ‘digest’ and ‘pass’ parts of the aforementioned idioms. Therefore, the mucosal immunity described previously, is also innervated by this nerve. Studies show that when vagus is dysfunctional, there is more GI inflammation and an overall dysregulated mucosal immunity.7,8 There is evidence for stress-induced alterations in gut flora and an associated increase in inflammatory markers in the gastrointestinal tract. Does it go both ways? If there is less stress, then there is less GI inflammation, but if there is more regulation of gut inflammation, increased intestinal integrity and a more diverse microbiome, will there be a more regulated stress response? Are we stimulating vagus by stimulating mucosal immunity, therefore eliciting a parasympathetic response? Is this actually the mechanism by which Lion’s Mane decreases depression and anxiety in post-menopausal women?9
Bidirectional gut-brain communication: One route of this communication is thought to begin through sensory information from the GI tract, and subsequent activation of neural, hormonal, and immunological signals. These signals can independently or cooperatively relay information to the central nervous system (CNS).10,11,12 There are a number of studies described in this review regarding increased probiotic intake associated with increased mood and less anxiousness. Specifically, probiotic supplementation with Lactobacillus helveticus and Bifidobacterium longum showed less self-reported negative mood and decreased urinary cortisol.13 A similar effect was also observed in healthy participants who consumed a mixture of Bifidobacterium bifidum and Bifidobacterium lactis, and Lactobacillus acidophilus, Brevibacillus brevis, Brevibacterium casei, Bifidobacterium salivarius, and Lactococcus lactis 14 Allen et al.15 found that healthy individuals fed Bifidobacterium longum had attenuated levels of cortisol and reduced subjective anxiety in response to the socially evaluated cold stress pressor test. It does indeed seem to go both ways: decreasing inflammation and regulating the gut microbiome reduces anxiety and stress, and reduced anxiety and stress decreases inflammation and regulates the gut microbiome.
Lion’s Mane mushroom is more than one constituent that increases nerve growth factor synthase (the myopia of this mushroom’s medicine). Lion’s Mane, like all things living, is made up of many synergistic compounds that work together to keep this mushroom living and producing. These compounds also happen to play a major role in human health. This field of neuroimmunology is growing and the physiological effects of Lion’s Mane are an excellent example of the mechanisms of bidirectional gut-brain communication. Lion’s Mane is, in fact, the ultimate psychosomatic medicine, in the true meaning of the word – relating to the interaction of mind and body, the psyche and the soma, and a true psychobiotic.
*Psychobiotic is actually a word, I did not invent it. It is a medicine that affects the psyche by regulating the gut microbiome.
Disclaimer: Information from this post is not meant to diagnose or treat any disease.
Work Cited
- Qin M, Geng Y, Lu Z, Xu H, Shi JS, Xu X, Xu ZH. Anti-Inflammatory Effects of Ethanol Extract of Lion’s Mane Medicinal Mushroom, Hericium erinaceus (Agaricomycetes), in Mice with Ulcerative Colitis. Int J Med Mushrooms. 2016;18(3):227-34. doi: 10.1615/IntJMedMushrooms.v18.i3.50. PubMed PMID: 27481156.
- Ren Y, Geng Y, Du Y, et al. Polysaccharide of Hericium erinaceus attenuates colitis in C57BL/6 mice via regulation of oxidative stress, inflammation-related signaling pathways and modulating the composition of the gut microbiota. J Nutr Biochem. 2018;57:67-76. doi:10.1016/j.jnutbio.2018.03.005
- Diling C, Xin Y, Chaoqun Z, et al. Extracts from Hericium erinaceus relieve inflammatory bowel disease by regulating immunity and gut microbiota. Oncotarget. 2017;8(49):85838-85857. doi:10.18632/oncotarget.20689
- Diling C, Chaoqun Z, Jian Y, et al. Immunomodulatory activities of a fungal protein extracted from Hericium erinaceus through regulating the gut microbiota. Front Immunol. 2017;8(JUN). doi:10.3389/fimmu.2017.00666
- Yang Y, Zhao C, Diao M, et al. The Prebiotic Activity of Simulated Gastric and Intestinal Digesta of Polysaccharides from the Hericium erinaceus. Molecules. 2018;23(12):1-14. doi:10.3390/molecules23123158
- Sheng X, Yan J, Meng Y, Kang Y, Han Z, Tai G, Zhou Y, Cheng H. Immunomodulatory effects of Hericium erinaceus derived polysaccharides are mediated by intestinal immunology. Food Funct. 2017 Mar 22;8(3):1020-1027. doi:10.1039/c7fo00071e. PubMed PMID: 28266682.
- Shea-Donohue T, Urban JF Jr. Neuroimmune Modulation of Gut Function. Handb Exp Pharmacol. 2017;239:247-267. doi: 10.1007/164_2016_109. Review. PubMed PMID: 28035531.
- Bailey MT. Psychological Stress, Immunity, and the Effects on Indigenous Microflora. Adv Exp Med Biol. 2016;874:225-46. doi: 10.1007/978-3-319-20215-0_11. Review. PubMed PMID: 26589222.
- Nagano M, Shimizu K, Kondo R, Hayashi C, Sato D, Kitagawa K, Ohnuki K. Reduction of depression and anxiety by 4 weeks Hericium erinaceus intake. Biomed Res. 2010 Aug;31(4):231-7. PubMed PMID: 20834180.
- Lach G, Schellekens H, Dinan TG, Cryan JF. Anxiety, Depression, and the Microbiome: A Role for Gut Peptides. Neurotherapeutics. 2018;15(1):36-59. doi:10.1007/s13311-017-0585-0
- Matteoli G, Boeckxstaens GE. The vagal innervation of the gut and immune homeostasis. Gut. 2013;62(8):1214-1222. doi:10.1136/gutjnl-2012-302550
- Fonseca RC, Bassi GS, Brito CC, et al. Vagus nerve regulates the phagocytic and secretory activity of resident macrophages in the liver. Brain Behav Immun. 2019;81(December 2018):444-454. doi:10.1016/j.bbi.2019.06.041
- Messaoudi M, Violle N, Bisson J-F, Desor D, Javelot H, Rougeot C. Beneficial psychological effects of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in healthy human volunteers. Gut Microbes 2011;2:256– 261.
- Steenbergen L, Sellaro R, van Hemert S, Bosch JA, Colzato LS. A randomized controlled trial to test the effect of multispecies probiotics on cognitive reactivity to sad mood. Brain Behav Immun 2015;48:258–264.
- Allen AP, HutchW, Borre YE, et al. Bifidobacterium longum 1714 as a translational psychobiotic: modulation of stress, electrophysiology and neurocognition in healthy volunteers. Transl Psychiatry 2016;6:e939.
Fall Transitions

Fall. The quintessential season of the mushroom. I very outwardly and not so secretly love when summer comes to an end. The giant ball of fire in the sky, reflecting off of the far too many reflective surfaces throughout the city like laser beams in a diamond shop, is finally going to be shrouded by clouds. What a treat. Although a creature of habit, I have a deep love for transition – seasons of transition, symbols of transition, momentous occasions initiating transition, etc. Perhaps this is why I love Fall, and perhaps this is why I have a tattoo of an ouroboros on my right arm, and most definitely why I love mushrooms. As the rains come and wash away the summer, this can be a time of deep introspection and with that, inevitable discomfort. Shorter days and a generally darker existence is not desirable to most people, but can be especially valuable for the psyche. So rather than seeking herbs or mushrooms to bring more ‘light’ during this time, how about seeking out plant and mushroom allies to support and encourage transition. I think of deciduous trees during this time, trees that innately hold the energy of transformation – the ability to move through seasons in different forms and continue to grow stronger each year. The tannins in the leaves of the Birch, Willow, Poplar and Aspen oxidize from green to brilliant yellow, red, and brown. They stand bare through winter – vulnerable, nude, yet with tremendous stature and strength. Imaginably, this is the medicine that these trees share, and the energy that we can cultivate. Look for patterns in nature to understand the medicine in nature. Is it grace through transition that you desire? Look closer at the natural world that surrounds you. This grace encompasses you already.
Oyster Mushroom
Pleurotus ostreatus
“side of the body” and “resembling oyster”
I am apt to write about the polypores – the often overlooked wizened and ancient ‘conks’ of the forest – that require more time to age, to collect, to process. I am stepping back and writing about the medicine of the familiar gilled Oyster Mushroom. They are as common in the forest as in the grocery store, and what better medicine to write about than medicine that is so accessible that the forest dwellers as well as the grocery store goers may benefit.
The fruiting Oysters will be found on hardwoods mostly – the Cottonwoods, Oaks and Alder – fruiting from their carnivorous mycelia in the spring. The carnivory is real – watch the mycelia digest a nematode in this video

Spore print
White
Known active constituents
Pleuran (polysaccharide), chrysin, ergothioneine, lovastatin, GABA
Nutritional information
Nutritional constituents estimated per 100 g dry weight include protein (29.3 g), carbohydrate (62.97 g), crude fat (0.91 g), ash (6.82 g) and crude fiber (12.3 g).
Energy value of this mushroom was about 297.5 kcal/100 g dry weight
Major mineral components estimated include Ca, Fe, and Mg with highest level of 505.0, 109.5 and 108.7 mg/100 g respectively.
Therapeutic actions
Immune modulating, antiatherogenic, neuroprotective, antioxidant, inflammation modulating
Some laboratory findings
Antiatherogenic
There are many factors that aggregate and result in atherosclerosis. These include and are not limited to hypertension, hyperglycemia, hypercholesterolemia and lipid peroxidation. Oyster mushroom research has demonstrated protection against all of those factors. In an animal study, rats were administered chrysin-rich Pleurotus extract. After administration there was a significant decrease in mean blood serum levels of glucose, lipid profile parameters, and hepatic marker enzymes and a simultaneous increase in enzymatic and nonenzymatic antioxidant parameters.2
P. ostreatus is the richest known source of ergothioneine, containing 118.91mg/kg. As mentioned previously, ergothioneine protects against DNA damage and lipid oxidation. For hypertension, Oyster mushroom water extract inhibits angiotensin converting enzyme (ACE) – a common mechanism in hypertensive medication.
Oyster mushroom also contains lovastatin, a naturally occurring statin compound that reduces LDL cholesterol through inhibition of HMG-CoA reductase.
Some human findings
Atopic dermatitis
In a split-body study of 80 patients, topical P. ostreatus-based β-glucan cream application resulted in improvement of both subjective and objective symptoms of atopic dermatitis. The patients applied the cream on one segment of the body with atopic dermatitis and no treatment on another atopic dermatitis segment. On the application site there was a significant decrease in the number of days and severity of atopic dermatitis. 4
You can make your own Oyster mushroom cream by following my mushroom cream recipe here. The aqueous part of the cream will be an Oyster mushroom decoction.
Respiratory Disease
Pleuran extract from Oyster mushrooms has clinical evidence for application with various respiratory diseases. In a human study, chronic obstructive pulmonary disease (COPD) patients treated with 100mg Pleuran, 60mg vitamin C, and 5mg Zinc, had significantly lower incidence and shorter duration of exacerbations compared to the control (60mg vitamin C and 5mg Zinc).5
In another double-blinded, placebo-controlled, randomized multicentric study, 175 children were treated with either pleuran or placebo over a 12-month period. Children treated with pleuran experienced a significant reduction in the frequency of recurrent respiratory tract infections. 1,6 These findings agreed with a Spanish study investigating 166 children aged one to ten years old who were also treated with Pleuran for recurrent respiratory infection.7
Advantageous respiratory effects of pleuran were also observed in adult athletes. A study included 50 athletes treated with pleuran over a three month period of time and found a significant reduction in the frequency of upper respiratory tract infections as compared to athletes treated with placebo. Blood samples of the athletes showed significantly higher levels of circulating NK cells in the pleuran group as compared to the placebo group.8
Antihyperlipidemic
20 subjects were randomized to take either one portion of soup containing 30g dried oyster mushrooms or a tomato soup (placebo) on a daily basis for 21 days. Standardized blood concentrations of lipid parameters and oxidized LDLs were measured at baseline and after 21 days. Treatment with Oyster mushroom soup decreased both triacylglycerol and oxidized low density lipoprotein levels significantly, and showed a significant tendency towards lowering total cholesterol values. 9
Pleurotus ostreatus Syrup Recipe
for upper respiratory support
Ingredients
30g dried Oyster mushrooms, chopped
120mL raw honey
130mL Water (up to 300mL if not using pressure cooker)
Kitchen Tools Needed
Boiling pot or Pressure cooker (Instant pot)
Cheese cloth
Potato ricer
Bowl
Directions
Pressure cooker: pressure cook dried mushrooms with 130mL water for 45 minutes
Boiling pot: boil for 1.5 hours, strain mushroom from decoction, dispose of mushrooms, put aqueous extract back on heat and reduce liquid down to 120mL
Once you have 120mL of mushroom aqueous extract, stir in 120mL of raw honey
Store in jar and refrigerate
Final product should be 240mL 1:8 (1gram of mushroom in 8mL liquid)
1g mushroom = about .5g Pleuran
Some math:
Pleurotus ostreatus is about 50% Polysaccharide, and the best way to extract these polysaccharides is through a boiling process. We have 30g of dried Pleurotus ostreatus and for a child who weighs 27kg, we would dose them with 270mg Pleuran, and so about 540mg dried oyster mushroom, or .5g. We want there to be .5g in 1 tsp (4mL) of extract. Since we will add 1part honey to 1part mushroom decoction, we will want the decoction to be a 1:4 and the final product to be a 1:8.
Pediatric Dose: 4mL (~.25g Pleuran)

Work Cited
- Jesenak M, Hrubisko M, Majtan J, Rennerova Z, Banovcin P. Anti-allergic Effect of Pleuran ( b -glucan from Pleurotus ostreatus ) in Children with Recurrent Respiratory Tract Infections. 2014;474(March 2013):471-474.
- Anandhi R, Annadurai T, Anitha TS, Muralidharan AR, Najmunnisha K, Nachiappan V, Thomas PA, Geraldine P. Antihypercholesterolemic and antioxidative effects of an extract of the oyster mushroom, Pleurotus ostreatus, and its major constituent, chrysin, in Triton WR-1339-induced hypercholesterolemic rats. J Physiol Biochem. 2013 Jun;69(2):313-23. doi: 10.1007/s13105-012-0215-6. Epub 2012 Oct 27. PubMed PMID: 23104078.
- Hamdi M, Abidin Z, Abdullah N, et al. Therapeutic properties of Pleurotus species ( oyster mushrooms ) for atherosclerosis : A review Therapeutic properties of Pleurotus species ( oyster mushrooms ). Int J Food Prop. 2017;20(6):1251-1261. doi:10.1080/10942912.2016.1210162.
- Jesenak M, Urbancek S, Majtan J, Banovcin P, Hercogova J. b -Glucan-based cream ( containing pleuran isolated from pleurotus ostreatus ) in supportive treatment of mild-to-moderate atopic dermatitis. 2016;6634(4):351-354. doi:10.3109/09546634.2015.1117565.
- Minov J, Bislimovska-karadzhinska J, Petrova T, Vasilevska K, Stoleski S. Effects of Pleuran ( Β – Glucan from Pleurotus Ostreatus ) Supplementation on Incidence and Duration of COPD Exacerbations. 2017;5(7):893-898.
- Jesenak M, Urbanclkova I, Banovcin P. Respiratory Tract Infections and the Role of Biologically Active Polysaccharides in Their. Nutrients. 2017:1-12. doi:10.3390/nu9070779.
- Pico Sirvent L, Sapena Grau J, Morera Ingles M, Rivero Urgell M. Effect of supplementation with β–glucan from Pleurotus ostreatus in children with recurrent respiratory infections. Ann Nurr Metab. 2013; 63 (1): 1378.
- Bergendiova K, Tibenska E. Pleuran ( b -glucan from Pleurotus ostreatus ) supplementation , cellular immune response and respiratory tract infections in athletes. 2011:2033-2040. doi:10.1007/s00421-011-1837-z.
- Schneider, I.; Kressel, G.; Meyer, A.; Krings, U.; Berger, R.G.; Hahn, A. Lipid Lowering Effects of Oyster Mushroom (Pleurotus Ostreatus) in Humans. Journal of Functional Foods 2011, 3(1), 17–24. DOI:http://Dx. Doi.Org/10.1016/J.Jff.2010.11.004
Ergothioneine: The Elusive Amino Acid

Proving that mushrooms are an essential part of the human diet
Definitions to consider while reading:
Antioxidant: Compounds that mitigate oxidation. Oxidation is a reaction that can produce free radicals, leading to cellular damage. Antioxidants such as thiols and ascorbic acid (vitamin C) terminate these reactions by donating electrons. To balance oxidative states, living organisms maintain systems with overlapping antioxidants such as glutathione, catalase, superoxide dismutase.
Blood-brain barrier: a filtering mechanism of the capillaries that carry blood to the brain and spinal cord tissue, blocking the passage of certain substances.
Cation chelator: a type of bonding of ions and molecules to metal ions. There is application of this in removing toxic metals from the body.
Glutathione: Glutathione is a tripeptide (cysteine, glycine, and glutamic acid) found in surprisingly high levels—5 millimolar—concentrations in most cells. Responsible for the following actions in the body: (Pizzorno)
- Direct chemical neutralization of singlet oxygen, hydroxyl radicals, and superoxide radicals
- Cofactor for several antioxidant enzymes
- Regeneration of vitamins C and E
- Neutralization of free radicals produced by Phase I liver metabolism of chemical toxins
- One of approximately 7 liver Phase II reactions, which conjugate the activated intermediates produced by Phase I to make them water soluble for excretion by the kidneys
- Transportation of mercury out of cells and the brain
- Regulation of cellular proliferation and apoptosis
- Vital to mitochondrial function and maintenance of mitochondrial DNA (mtDNA)
Lipid peroxidation: The oxidative degradation of lipids. It is the process in which free radicals “steal” electrons from the lipids in cell membranes, resulting in cell damage. (Wik)
Oxidative stress: disturbance in the balance between the production of reactive oxygen species (free radicals) and antioxidant defenses (Betteridge)

‘Constituentism’
I am not necessarily a fan of isolation and ‘constituentism’ in supplemental medicine – especially when a constituent has been isolated from its synergistic community of molecules from the whole plant or whole mushroom and is considered supposedly different than a pharmaceutical. Though, I do love to understand the individual constituents in organisms and how they work independently, in order to better understand how they work synergistically. There is also a greater understanding to be had of how animals, plants, fungi and bacteria all evolved together when we can explore specific plant and fungi constituent mechanisms with specific cellular receptors and transporters in the animal organism. A specific example of this is a compound called Ergothioneine (ERG). ERG is an amino acid derivative, specifically a crystalline betaine, derivative of histidine – the etymology explains; ergot- (found in ergot (fungus) thione- (double bond to a Sulphur) and -ine (amine containing)—basically a sulphur containing amine found in fungi. ERG is also found in Actinobacteria (gram positive bacteria that behave similar to fungi in soil, helping to decompose the organic matter of dead organisms so the molecules can be taken up by new plants, they even grow extensive mycelium like fungi and were in fact long believed to be fungi) and Cyanobacteria, commonly known as blue-green algea, (another abnormal bacteria that can actually obtain their energy from photosynthesis – they are the only photosynthetic prokaryote able to produce oxygen). (blankenfeldt)
Specific ERG transporters in the animal
We have evolved so closely with mushrooms that we have specific transporters and receptors uniquely for mushroom compounds. ERG is no exception. While we have a specific receptor for ERG, we cannot synthesize this molecule ourselves, and as I will describe later, we are discovering this compound is extremely important for human health. The ERG transporter, OCTN1, (gene: SLC22A4) is found specifically on erythrocytes, fetal liver and bone marrow, ileum of the small intestine, trachea, kidney, cerebellum, lung, monocytes, seminal vesicles and the lens and cornea of the eye (Halliwell). Interestingly, ERG transporter is concentrated in the mitochondria of cells – suggesting a role in protecting mitochondrial components from DNA damage (Paul, Halliwell). This transporter’s only known role is to sequester as much ERG within the cells as is available, and only cells with this transporter can absorb, distribute, and retain this compound (Grundermann). Researchers explored the effects when cells were depleted of this transporter, the results being that cells were more susceptible to oxidative stress, leading to mitochondrial damage, protein oxidation, and lipid peroxidation. Once taken up into the cell, ERG is extremely bioavailable and is retained for up to a month within the body.

Bioavailability of ergothioneine from mushroom consumption
Once consumed in the diet, whether it be the isolated molecule or from whole mushrooms, ERG is quickly absorbed into the blood stream. Human erythrocytes (red blood cells, RBC) contain 2-9 fold more ERG than plasma. Interestingly, concentration is lowest in early life (1-10yo), increases between 11-18yo and reaches maximum value of 3.7mg/100mL by age 18. In this study, subjects consumed 8g and 16g of mushrooms and increases in red blood cell ERG were observed. After 1 and 4hr of consumption, the 16g mushroom dose increased RBC ERG concentration compared to the control, and after only 2 hrs of 16g of mushroom consumption, RBC ERG concentration was significantly higher than the control. Variability in bioavailablity was due to genetic variations in the SLC22A4 gene (Heller, Kris-etherton, Beelman). Interestingly, mutations in the ERG transporter have been identified as a susceptibility factor for autoimmune disorders, such as rheumatoid arthritis and Crohn’s disease, as well as neurodegenerative disorders, such as dementia and Parkinson’s (Paul, Snyder).

Why do we care – Once ergothioneine is in the cell, then what happens?
Ergothioneine as an antioxidant and cytoprotectant: The distribution of ERG transporters may seem random, but on closer inspection, they seem to be present in tissues predisposed to high levels of oxidative stress and inflammation (Halliwell). In animal studies, animals who were completely deficient in ERG had higher levels of reactive oxygen species and were therefore more susceptible to oxidative stress. In vitro, ERG is a powerful scavenger of hydroxyl radical and has been shown to deactivate singlet oxygen at a higher rate than glutathione (Hseu, Servillo). Similar results were proven in vivo – rats supplemented with ERG had lower levels of lipid peroxidation and higher levels of glutathione and alpha-tocopherol. As one might surmise, ERG and glutathione seem to have an intimate relationship within the cell. Glutathione is considered the major intracellular antioxidant in almost all organisms and has important functions in detoxification and immune function. It has been proposed, based on current research, that ERG can help maintain GSH levels in the presence of oxidative burden by interacting with other cellular defense systems. The maintenance of glutathione tissue levels is important in maintaining health as depletion will impair immune function. Conveniently, mushrooms contain both glutathione and ERG. In fact, mushrooms have been observed to have higher GSH amounts than any vegetable or fruit. Grifola frondosa (Maitake), Hericium erinaceus (Lions mane), Pleurotus ostreatus (Oyster mushroom), Boletus edulis (porcini) and Lentinus edodes (shitake) containing the most, respectively (Kalaras). Considering the relationship between ERG and GSH in mushrooms, a correlation analysis found that mushrooms high in GSH were also high in ERG, specifically the caps or pileus of the mushrooms (kalaras). All this said, mushrooms are an important source for cellular antioxidants.
Cation chelator: ERG chelates divalent metal cations – specifically, Cu2, hg, Zn, Cd, Co, Fe, and Ni (Cheah, Song, Kerley). Binding these cations in the body may help prevent their participation in the generation of reactive oxygen species. For example, EGT has been found to protect DNA and protein against copper induced oxidative damage through formation of a redox-inactive EGT-Cu complex. Interestingly, the high levels of ERG in semen – due to the high concentration of ERG OCTN1 transporter on the seminal vesicles- have been shown to prevent Cu inhibition of sperm motility. There is more to be explored between the relationship of ERG and semen viability.
Ergothioneine, aging, and cognitive decline: As we age, we are more vulnerable to the oxidative stress and environmental toxins that slowly damage our DNA, making us more susceptible to neurodegeneration – one of many conditions associated with DNA damage from oxidative stress. Low levels of glutathione have been linked to certain neurodegenerative diseases including Parkinson’s disease (Wei, Shah). ERG was able to dose dependently enhance glutathione activity in the rat liver cytosol—leading researchers to postulate that declining ERG may play a role in age related decline of GSH and glutathione peroxidase. Furthermore, ERG levels were found to be lower in the elderly with early stages of dementia and in PD patients relative to age matched healthy controls (Cheah). In animal studies, oral administration of ERG protected neurons and preserved cognitive function following administration of toxic amyloid beta cisplatin or D-galactose. It is now understood that the presence of Octn1 transporter in the blood brain barrier is responsible for these neuroprotective actions. Captivatingly, researchers found that there is a direct relationship between whole blood and brain ERG levels following consumption (Cheah). It is rare for compounds to be bioavailable in this way and transported across the blood brain barrier so readily – even glutathione needs to be taken intranasal for treatment of neurodegeneration.
Ergothioneine human trial: uptake metabolism and effects
While there is undeniably a lack of human trials exploring the in vivo effects of ERG, a recent study from 2017 explored ERG uptake, metabolism, and effects on biomarkers of oxidative damage and inflammation in healthy human subjects (cheah, Tang). One of the most interesting aspects of ERG discussed in this particular article is that ERG is a tautomer. This means that it exists in two forms – thione and thiol. The thiol is a single bond to sulfur and the thione is a double bond to sulfur. The article explains that in animal physiologic conditions, ERG primarily exists as the thione tautomer – under circumstances of low stress in the body, ERG remains in its thione tautomer form and is not the first choice as an antioxidant, rather endogenous antioxidants like glutathione are preferred (glutathione is a primary antioxidant thiol in the body). When the cells undergo higher levels of oxidative stress, ERG transforms into its thiol tautomer form and is then used for extra support. Additionally, under levels of elevated stress, tissues have increased amounts of the ERG, supposedly by upregulating expression of the OCTN1 ERG transporter in response to inflammatory cytokines.
This study also found that ERG can be stored in the cells for up to a month. It is theorized that ERG is stored for an extended period of time following consumption until it is required as a stronger defense mechanism.
During administration of ERG, plasma levels significantly elevated, while whole blood levels steadily increased for up to four weeks after administration stopped (red blood cells could continue to take ERG up as needed), and the excretion of ERG in the urine remained low, indicating that ERG is absorbed and retained in the body after oral administration. While this study used pure ERG, other studies previously mentioned (Heller) prove this same high bioavailability of ERG from dietary mushrooms, and so I will consider this as true for dietary ERG.

Mushroom analyses of ergothioneine: Fruiting body or mycelium? Gilled mushrooms or polypore mushrooms?
ERG was highest in the fruiting body of Pleurotus ostreatus and in the mycelium of Pleurotus eryngii. Generally, fruiting bodies and mycelium contained different amounts of ERG, with Pleurotus genus containing the highest amounts overall (Chen). Among simple mushrooms, fruiting body of White Button had the least (1.4mg erg per 85g mushroom) and Portabella the highest (2.7mg erg per 85g mushroom). While among specialty mushrooms, Maitake had the least (16.3mg erg per 85g mushroom) and oyster the highest (26.4mg per 85g mushroom) (Dubost).
Fruiting bodies of gilled mushrooms, specifically the cap, contain the most ERG, while polypore mushrooms contain very small amounts. Interestingly, the mycelium of Ganoderma contains more ERG than the fruiting body, but still not as much as the fruiting bodies of Shitake, Matsutake, Oyster, and Maitake (Lee, Wang).
Extraction:
Hot water extraction is sufficient to extract ERG from mushrooms.
In a study that explored water extraction – temperature of water, ratio of water to mycelium and the extraction rate of ERG, the following information was found:
Most notable: at 85°C and 20:1 ratio of mycelium to water, the extraction rate of ERG was 91.2% – the least effective yield of ERG was at 78°C and a 20:1 ratio of mycelium to water. There was no difference in ERG concentration between 5 minutes and 120 minutes, so a long boil to extract EGR is not necessary (Zhang).
In Summary:
Ergothioneine is a water soluble compound that is most abundant in Oyster mushrooms. There are transporters on different tissues in the body that are highly specific to ergothioneine. Ergothioneine is readily absorbed into the blood after consumption of mushrooms and stored in tissues for up to 1 month. In times of excessive oxidative stress, ergothioneine is taken up by those tissues and used as an antioxidant. Of note: there are transporters on the blood brain barrier and there is an association with low ergothioneine and age-related cognitive decline. In a world full of environmental toxins that are mostly impossible to escape, we might as well eat more mushrooms and get some extra protection.
How to apply this information to your life:
Eat a lot of mushrooms, especially Oyster mushrooms.
Oyster Mushroom Recipes
Resources for ergothioneine rich mushrooms around the Seattle area:
Work Cited
Antonicelli F, Aruoma OI. Ergothioneine inhibits oxidative stress- and TNF- a -induced NF- j B activation and interleukin-8 release in alveolar epithelial cells. 2003;302:860-864. doi:10.1016/S0006-291X(03)00224-9.
Aruoma OI, Spencer JPE, Mahmood N. Protection Against Oxidative Damage and Cell Death by the Natural Antioxidant Ergothioneine. 1999;37.
Aruoma OI, Whiteman M, England TG, Halliwell B. Antioxidant Action of Ergothioneine : Assessment of Its Ability to Scavenge Peroxynitrite. 1997;391(231):389-391.
Aubert DIRKT. Dietary Sources and Antioxidant Effects of Ergothioneine. 2007:6466-6474. doi:10.1021/jf071328f.
Benson KF, Ager DM, Landes B, Aruoma OI, Jensen GS. Improvement of joint range of motion ( ROM ) and reduction of chronic pain after consumption of an ergothioneine-containing nutritional supplement. Prev Med (Baltim). 2018;54(2012):S83-S89. doi:10.1016/j.ypmed.2012.02.001.
Betteridge, JD What Is Oxidative Stress? 2000:3-8.
Blankenfeldt W, Seebeck FP. Ergothioneine Biosynthetic Methyltransferase EgtD Reveals the Structural Basis of Aromatic Amino Acid Betaine Biosynthesis. 2015:119-125. doi:10.1002/cbic.201402522.
Cheah IK, Halliwell B. Biochimica et Biophysica Acta Ergothioneine ; antioxidant potential , physiological function and role in disease ☆. BBA – Mol Basis Dis. 2012;1822(5):784-793. doi:10.1016/j.bbadis.2011.09.017.
Chen S, Ho K, Hsieh Y, Wang L, Mau J. LWT – Food Science and Technology Contents of lovastatin , g -aminobutyric acid and ergothioneine in mushroom fruiting bodies and mycelia. LWT – Food Sci Technol. 2012;47(2):274-278. doi:10.1016/j.lwt.2012.01.019.
Deiana M, Rosa A, Casu V, et al. modulates oxidative damage in the kidney and liver of rats in vivo : studies upon the profile of polyunsaturated fatty acids L -Ergothioneine. 2004;5614:183-193. doi:10.1016/S0261-5614(03)00108-0.
Dubost NJ, Ou B, Beelman RB. Food Chemistry Quantification of polyphenols and ergothioneine in cultivated mushrooms and correlation to total antioxidant capacity. 2007;105:727-735. doi:10.1016/j.foodchem.2007.01.030.
Franzoni F, Colognato R, Galetta F, et al. An in vitro study on the free radical scavenging capacity of ergothioneine : comparison with reduced glutathione , uric acid and trolox. 2006;60:453-457. doi:10.1016/j.biopha.2006.07.015.
Gruber J, Fong S, Chen C, et al. Mitochondria-targeted antioxidants and metabolic modulators as pharmacological interventions to slow ageing. Biotechnol Adv. 2013;31(5):563-592. doi:10.1016/j.biotechadv.2012.09.005.
Gründemann D. The ergothioneine transporter controls and indicates ergothioneine activity — A review. Prev Med (Baltim). 2018;54(2012):S71-S74. doi:10.1016/j.ypmed.2011.12.001.
Halliwell B, Cheah IK, Drum CL. Biochemical and Biophysical Research Communications Ergothioneine , an adaptive antioxidant for the protection of injured tissues ? A hypothesis. Biochem Biophys Res Commun. 2016;470(2):245-250. doi:10.1016/j.bbrc.2015.12.124.
Harlfinger S, Golz S, Geerts A, et al. Discovery of the ergothioneine transporter. 2005.
Hseu Y, Lo H, Korivi M, Tsai Y, Tang M. Free Radical Biology and Medicine Dermato-protective properties of ergothioneine through induction of Nrf2 / ARE-mediated antioxidant genes in UVA-irradiated Human keratinocytes. Free Radic Biol Med. 2015;86(91):102-117. doi:10.1016/j.freeradbiomed.2015.05.026.
Kalaras MD, Richie JP, Calcagnotto A, Beelman RB. Mushrooms: A rich source of the antioxidants ergothioneine and glutathione. Food Chem. 2017;233:429-433. doi:10.1016/j.foodchem.2017.04.109.
Kerley RN, Mccarthy C, Kell DB, Kenny LC. Free Radical Biology and Medicine The potential therapeutic e ff ects of ergothioneine in pre-eclampsia. 2018;117(August 2017):145-157. doi:10.1016/j.freeradbiomed.2017.12.030.
Lee WY, Park E-J, Ahn JK, Ka K-H. Ergothioneine Contents in Fruiting Bodies and Their Enhancement in Mycelial Cultures by the Addition of Methionine. Mycobiology. 2009;37(1):43. doi:10.4489/MYCO.2009.37.1.043.
Lo Y, Lin S, Ulziijargal E, et al. Comparative Study of Contents of Several Bioactive Components in Fruiting Bodies and Mycelia of Culinary-Medicinal Mushrooms. 2012;14(4):357-363.
Li RWS, Yang C, Sit ASM, et al. Uptake and Protective Effects of Ergothioneine in Human Endothelial Cells. 2014;(September):691-700.
Markova NG, Karaman-jurukovska N, Dong KK, Damaghi N, Smiles KA, Yarosh DB. Free Radical Biology & Medicine Skin cells and tissue are capable of using L -ergothioneine as an integral component of their antioxidant defense system. Free Radic Biol Med. 2009;46(8):1168-1176. doi:10.1016/j.freeradbiomed.2009.01.021.
Martin KR. The Bioactive Agent Ergothioneine, a Key Component of Dietary Mushrooms, Inhibits Monocyte Binding to Endothelial Cells Characteristic of Early Cardiovascular Disease. 2010;13(6):1340-1346.
Moncaster JA, Walsh DT, Gentleman SM, Jen L, Aruoma OI. Ergothioneine treatment protects neurons against N -methyl- d – aspartate excitotoxicity in an in vivo rat retinal model. 2002;328:55-59.
Nakamichi N, Nakayama K, Ishimoto T, Masuo Y. Food-derived hydrophilic antioxidant ergothioneine is distributed to the brain and exerts antidepressant effect in mice. 2016;477:1-10. doi:10.1002/brb3.477.
Nguyen TH, Giri A, Ohshima T. A rapid HPLC post-column reaction analysis for the quantification of ergothioneine in edible mushrooms and in animals fed a diet supplemented with extracts from the processing waste of cultivated mushrooms. Food Chem. 2012;133(2):585-591. doi:10.1016/j.foodchem.2012.01.061.
Onofrio ND, Servillo L, Giovane A, et al. Free Radical Biology and Medicine Ergothioneine oxidation in the protection against high-glucose induced endothelial senescence : Involvement of SIRT1 and SIRT6. Free Radic Biol Med. 2016;96:211-222. doi:10.1016/j.freeradbiomed.2016.04.013.
Paul, BD, Snyder, SH. The Unusual Amino Acid, l-ergothioneine is a Physiologic Cytoprotectant. NIH public access 2010: 17-7. Doi: 10.1038/cdd.2009.163
Pizzorno, J., N.D. (2014). Glutathione! Integrative Medicine, 13(1), 8-12. Retrieved from https://search-proquest-com.buproxy.bastyr.edu/docview/1504261168?accountid=1173
Sakrak O, Kerem M, Bedirli A, et al. Ergothioneine Modulates Proinflammatory Cytokines and Heat Shock Protein 70 in Mesenteric Ischemia and Reperfusion Injury. 2018;42(2008):36-42. doi:10.1016/j.jss.2007.04.020.
Servillo L, Castaldo D, Casale R, et al. Free Radical Biology and Medicine An uncommon redox behavior sheds light on the cellular antioxidant properties of ergothioneine. Free Radic Biol Med. 2015;79:228-236. doi:10.1016/j.freeradbiomed.2014.11.017.
Shah SP, Duda JE. Dietary modifications in Parkinson ’ s disease : A neuroprotective intervention ? Med Hypotheses. 2018;85(6):1002-1005. doi:10.1016/j.mehy.2015.08.018.
Song T, Chen C, Liao J, Ou H, Tsai M. Ergothioneine protects against neuronal injury induced by cisplatin both in vitro and in vivo. Food Chem Toxicol. 2010;48(12):3492-3499. doi:10.1016/j.fct.2010.09.030.
Tomomi I, Manami K, Hironobu T, Etsuko H, Toshio N, Toshihiko O. Ergothioneine as an Anti-Oxidative / Anti-Inflammatory Component in Several Edible Mushrooms. 2011;17(2):103-110.
Wang X, Zhang J, Wu L, et al. A mini-review of chemical composition and nutritional value of edible wild-grown mushroom from China. Food Chem. 2014;151:279-285. doi:10.1016/j.foodchem.2013.11.062.
Weigand-heller AJ, Kris-etherton PM, Beelman RB. The bioavailability of ergothioneine from mushrooms ( Agaricus bisporus ) and the acute effects on antioxidant capacity and biomarkers of in fl ammation. Prev Med (Baltim). 2018;54(2012):S75-S78. doi:10.1016/j.ypmed.2011.12.028.
Wei Z, Li X, Li X, Liu Q, Cheng Y. Oxidative Stress in Parkinson ’ s Disease : A Systematic Review and. 2018;11(July):1-7. doi:10.3389/fnmol.2018.00236.
Yoshida S, Shime H, Funami K, Takaki H. The Anti-Oxidant Ergothioneine Augments the Immunomodulatory Function of TLR Agonists by Direct Action on Macrophages. 2017:1-15. doi:10.1371/journal.pone.0169360.
Zhang W, Liu Q, Zhou T, Mei B, Chen N, Jiang W. Aqueous Extraction of Ergothioneine from Mycelia of Pleurotus ostreatus and Ergothioneine Accumulation Regularity during Submerged Fermentation. Res Rev J Microbiol Biotechnol. 2016;5(1):1-8. https://www.rroij.com/open-access/aqueous-extraction-of-ergothioneine-from-mycelia-of-pleurotusostreatus-and-ergothioneine-accumulation-regularity-duringsubmerged-f-.pdf.
Mycophysiology and Hominid Perks

Take a moment to forget.
Forget about the idea that mushrooms have chemicals that have pharmacological potential. Forget that you want to learn about the mushrooms so that you can ‘use’ them.
Just for a moment, let’s see the mushrooms as their own organism.
Their own organism with all of their chemicals not for the human system, but for the fungal system.
Every constituent that we wish to extract and isolate and identify has an important role within the living mushroom, and a role within the entire ecosystem.
Let’s explore these compounds and their functions within the mushroom before exploring their functions and relations within the human system.
Remember, a human is not their cholesterol, a human is not their pheromones, a human is not their amino acids. A human is a synergistic organism made up of thousands of molecules and cells all working together. It is impossible to understand who a human is from just analyzing one of these molecules.
When we look at the mushroom through the lens of its parts, keep in mind it is the synergism between these parts that makes the organism, not the isolated constituents.
The purpose of this writing is to first investigate major myco-constituents that have been researched for their medicinal value and understand their innate role in the fungal organism. After delving into the mushrooms on a cellular level I’ll discuss how these constituents interact with the human system. We did after all evolve with the mushrooms, we just work so beautifully together. Through understanding the innate role of these compounds and where they are within the mushroom organism, we gain insight into the best ways to extract these compounds to support our own physiology. Specific constituents that will be discussed: Polysaccharides, volatile compounds (Alcohols, monoterpenes, sesquiterpenes, aldehydes, ketones), triterpenes, ergosterol, phenolic compounds, and fatty acids.
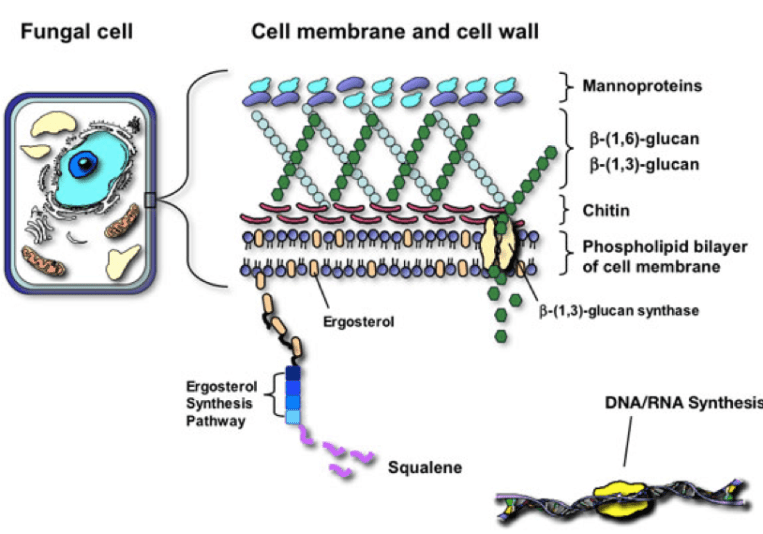
The Fungal Cell
The cell wall is an exquisite and imperative structure in all organisms. The cell wall role in fungi is similar to the role of the cell membrane in humans – it is dynamic in controlling the shape and protecting the organism from the environment. The cell well is made up of compounds involved in morphogenesis, reproduction, cell-cell and cell-matrix interaction. The fungal cell is rigid as it is made up of one of the hardest substances, chitin. Although rigid, it must also be complex to allow for budding, growth and adaptation to environmental stress. The fungal cell wall is composed of multidimensional, communicating polysaccharides including chitin and glucans. (Taborda)
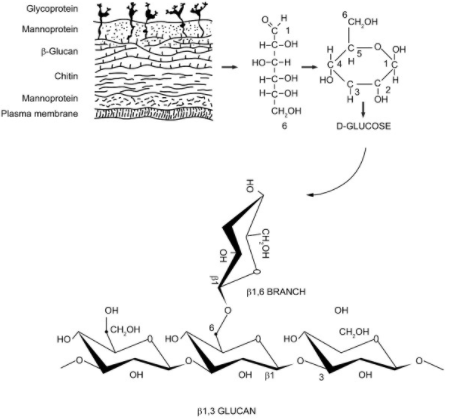
Polysaccharides
– Many simple sugar molecules attached to one another, referred to as glucans when it is glucose molecules-
In the Mushroom
These chains of sugars may be extracellular (outside the cell wall), may be associated with the cell wall and membrane, or may be intracellular. Extracellular polysaccharide function is unclear, pullulan a polysaccharide similar to glycogen and mycodextran are alpha linked glucans that are extracellular. From what I could gather, the function of these may be as storage reserves. Beta 1,3 and beta 1,6 glucans are also sometimes found on the outside of the cell, but are most often important components of the cell wall itself. (Griffin) The major role of polysaccharides is in providing cell wall structure. As Basidiomycota (fungi with fruiting bodies) grow and mature fruiting bodies, the total polysaccharide levels increase. (Friedman)
With this knowledge about the role of these compounds as structural support and barrier to the fungal cell we can understand the role within the human organism.
In the Human
The polysaccharides act in the human in an analogous way as they act in the fungal cell. They support the human cells in responding to environmental stressors within the human terrain.
Polysaccharides play a number of functions in the human system. Once ingested, these compounds move through the GI, coming into contact to mucous membranes all the way down through the inner tube from mouth to anus. The mucous membranes have lymphoid tissue called gut associated lymphoid tissue (GALT) that contains immune cells called macrophages. These macrophages, or big eaters, are an important part of our innate immune system. These immune cells have specific receptors for beta-glucans, Dectin-1 and TLR2. Once the beta- glucans come into contact with them, there are chemical messengers called cytokines that are stimulated. Specifically, IL-12 and IFN-y. These cytokines are important in stimulating Natural killer cells, Cytotoxic T cells and TH1 cells. Once these cells are stimulated they target viruses and cancer cells. (guggenhein)
Polysaccharides also function as soluble fiber. When they enter the gut, they slow the release of glucose into the blood stream, acting as hypoglycemic compounds. They also help to decrease cholesterol through binding bile acids. When bile acids end up in the soluble fiber, they are pooped out and then the body will use cholesterol to make more, rather than recycling the bile acids. Along with binding bile acids, they also prevent absorption of cholesterol from food in the GI, further lowering serum cholesterol. These polysaccharides are also beneficial in controlling blood sugar. Polysaccharides inhibit the enzyme, alpha-glucosidase, this enzyme is known to induce after meal hyperglycemia. Along with inhibiting this enzyme, polysaccharides have also been shown to upregulate the GLUT 4, insulin-response glucose transporter, while down regulating NFkappa-B, a nuclear transcription factor that controls regulation of inflammation. (friedman, shamtsyan)
Polysaccharides as antioxidants: Polysaccharides increase activities of liver oxidative enzymes, catalase, glutathione peroxidase, and superoxide dismutase and increase glutathione and malondialdehyde levels. This is all to say that these compounds support our innate antioxidant systems supporting the human in relieving oxidative stress and free radical damage.
Polysaccharides as prebiotics: The oligosaccharides and polysaccharides contained in mushrooms are consumed by Bifidobacterium and Lactobacilli, two important bacteria in a healthy microbiome. Through supporting a healthy gut microbiome, this can also be helpful with asthma, allergies, anxiety and even depression.
Extraction method:
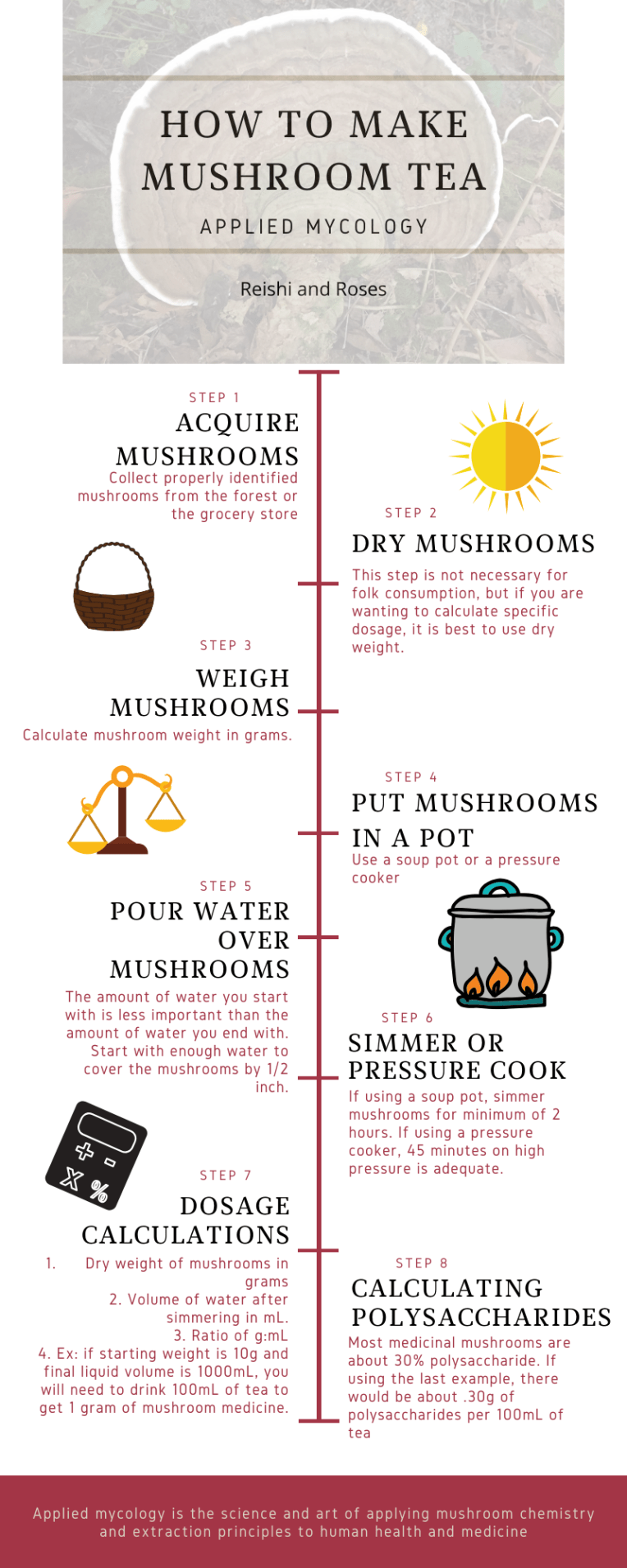
Hot water – 100 degrees C for at least 2 hours
Volatiles Compounds and Triterpenes
Low molecular weight, carbon based compounds that vaporize at about 20 degrees Celsius. Include: Alcohols, monoterpenes, sesquiterpenes, aldehydes, and ketones
Triterpenes have 30 carbons and are too heavy to be volatile, but I put them here since they are made in the same pathway as monoterpenes and sesquiterpenes.

In the Mushroom
These volatile compounds are derived from primary and secondary metabolism pathways; they diffuse through the atmosphere as “infochemicals”
These chemicals function as attractants and deterrents to insects and other invertebrates. Fungal volatiles have important properties as pheromones and defense. Some of these compounds are an efficient way of defending against fungal feeders. Octanol, often called mushroom alcohol, is an alcohol that deters banana slugs from eating mushrooms while also serving as an attractant for fungus eating beetles. Amusingly, octanol from human skin serves as a host odor cue that attracts blood sucking insects. Trametes versicolor is an example of a polypore mushroom that produces sesquiterpenes: specifically, cardinene, beta guiaene, isoledene and gamma patchoulene that attract fungivorous beetles. (morath,Rosecke) Through attracting these insects the mushrooms are better able to spread their spores and inoculate nearby substrate. A good way to make more of themselves!
Monoterpenes, sesquiterpenes and triterpenes are all compounds in mushrooms that have a cholesterol backbone. (The chemical structure looks similar to cholesterol)
Triterpenes are the heaviest of the three since they have 30 carbons and the others have 10 and 15 carbons respectively. Within the mushroom cell, triterpenes are converted into various metabolites including sterols, steroids, and saponins. They are produced via a pathway called the mevalonate pathway with a starting molecule called acetyl-coa. Animals have a very similar pathway that leads to cholesterol synthesis! This pathway in fungi also leads to ergosterol (a precursor to vitamin D2) which plays the same role that cholesterol plays in the human cell. It is a major component of the plasma cell membrane in fungi, important for membrane permeability.
Cytochrome p450 is important in gene expression and triterpenoid production. There is significantly more CYP450 during the transition from primordia to fruiting bodies, correlating with a higher triterpene content in the mushroom fruiting bodies. – developmental stage of fungal growth plays an important role in the regulation of secondary metabolite genes. (rep NP) (Schmidt-dannert, Schrader)
In the Human
Volatile compounds are very low molecular weight and cross easily through barriers and membranes. It is for this reason that essential oils are so readily absorbed through the skin and mucous membranes. Some volatile compounds can be neurotoxic for this reason, and some can be neuroprotective. There is research now exploring the role of octanol as an antiseizure agent. (Manjarrez-Marmolejo, chang, jahromi) Octanol reduced seizure induction and seizure discharges when it was applied directly to the epileptic focus in the somatosensory cortex. Additionally, it has been reported that administration of octanol significantly reduces the frequency and amplitude of epileptiform spikes, as well as the epileptic behavioral score induced by the administration of penicillin. (Manjarrez).
Some fungal sesquiterpenes have also been shown to inhibit TGF-b, decreasing kidney fibrosis in late stage kidney disease. Triterpenes are major compounds of interest in cancer research. Triterpenes have been shown to be directly cytotoxic to many cancer cell lines. (Rios, Grienke, Bhattarai, Ren, Jin, , Zhu) Triterpenes also have specific antiviral activity, inhibiting an enzyme, neuraminidase, which is important for viruses to be able to leave one cell to enter another. (Teplyakova, Gao) Triterpene compounds have been shown to be very beneficial in treatment of hypersensitivity reactions. Hypersensitivity reactions include allergy, asthma, dermatitis, and rhinitis. Triterpenes inhibit histamine release from mast cells – less histamine will result in less of a reaction. These can also be used in treatment of inflammatory reactions caused by insect stings and bites. (Rios)
Extraction Method
Volatile compounds need to be extracted from fresh mushrooms and will escape into the atmosphere with heat. They are alcohol and fat soluble. Best to do a cold ethanol extraction for volatile compounds. 24 hours is sufficient.
Triterpenes will not volatilize and are also fat and alcohol soluble. There are terpenes within the cell, and so it is best to break the chitin cell wall first with a water decoction, before proceeding with ethanol to extract the triterpenes. A triple extraction method will be provided at the end of this post.
Fatty acids
– Molecules that are long chains of lipid-carboxylic acid found in fats and oils and in cell membranes as a component of phospholipids and glycolipids –
In the Mushroom
Principal lipids in mushrooms are palmitic and linoleic acid. The fruiting body and pileus (cap) contains more linoleic acid and the stipe contains for oleic acid. The linoleic acid plays a role in fungal reproductive mechanisms. Trametes versicolor contains more short chain fatty acids – octanoic, decanoic and lauric acid. (Summer) Fungal membranes contain large quantities of free fatty acids, specifically glycerolipids and acylglycerols. These fatty acids occur in fungi as the major constituents of oil droplets suspended in mycelial and spore cytoplasm. They are also minor constituents of membranes and cell walls. Their primary function is storage material. Phophoglycerides are formed from a combination of fatty acids and are found primarily in the plasma membrane of cellular organelles, where they occur as complexes with proteins.
In the Human
Short chain fatty acids found in Trametes species modifies human fecal microbiota composition, increasing healthy bacteria – Bifidiobacteria and Lactobacillus while decreasing E.coli. (Yu) Short chain fatty acids also lower the intestinal pH, inhibiting growth of microbial pathogens (Plummer)
These fatty acid compounds also have antioxidant activities found to be as effective as alpha-tocopherol (Vit E) while also showing significant antimicrobial and anti-fungal activity. (Younis)
Extraction method:
Fat and alcohol soluble
Phenolic compounds
-The term ‘phenolic’ or ‘polyphenol’ can be defined chemically as a substance which possesses an aromatic ring bearing one or more hydroxy substituents, including functional derivatives (esters, methyl ethers, glycosides etc.)- Commonly known phenolic compounds include: bioflavonoids and proanthocyanidins
In the Mushroom
The phenolic compounds in mushrooms are secondary metabolites derived from intermediates of the shikimic acid pathway, the primary role of which is to provide the essential aromatic amino acids phenylananine, tyrosine and tryptophan. The intermediates of the shikimic acid pathway are precursors of aromatic compounds, including phenolic compounds. The biosynthesis of these compounds has demonstrated that they possess enzymes such as ammonia-lyases that convert phenalynine and tyrosine to cinnamic acids. These compounds represent the building blocks for pigments in mushrooms. The phenolic compound hispolon is an example of a yellow pigment in mushrooms. (Velisek)
In the Human
Phenolic compounds act as powerful antioxidants in the human organism. These compounds help to reduce oxidative stress through their own free radical scavenging activity as well as through stimulating the innate human antioxidant systems. (Geng) Phenolic compounds in mushrooms have also been shown to inhibit alpha-glucosidase in rats and significantly help with diabetic complications caused by this compound. A specific phenolic compound, hispolon, found in Phellinus species has been shown to have analgesic and anti-inflammatory effects as well as inhibits the growth of human cancer cells via the inhibition of the cytokine, TGF-beta. Hispolon also activates caspase, an important enzyme that induces cancer cell death. (Govindappa, Huang, Hong, Chang, Wu, Hsieh)
Extraction Method:
Ethanol or Water depending on how polar the phenolic compound is, best to do a triple extraction so you are sure to get them all.
Triple Extraction
The best way to get all of these compounds in one extract is to do a triple extraction method. The first part of the triple extraction is an overnight ethanol extraction. I use 95% ETOH and poor it over finely chopped/shredded fresh mushroom. Let this sit for 24-48 hours. Press the ETOH from the mushroom material and set aside (ETOH extract #1). Next, place the mushrooms from the original extraction into a crockpot or soup pot, cover with water and simmer for 2-12 hrs (The aqueous extract). Next, place these mushrooms and aqueous extract into a jar and leave the jar 1/3 empty. Fill the last 1/3 of the jar with the ETOH extract #1. Let sit (macerate) for a few weeks, shaking and loving daily. Press out mushrooms (the marc) from ETOH/Aqueous solvent (menstruum). Now you have your triple extraction – containing the volatile compounds, the polysaccharides, the triterpenes, phenolic compounds, and fatty acids.
Work Cited
- Bhattarai G, Lee Y-H, Lee N-H, et al. Fomitoside-K from Fomitopsis nigra Induces Apoptosis of Human Oral Squamous Cell Carcinomas (YD-10B) via Mitochondrial Signaling Pathway. Biol Pharm Bull. 2012;35(10):1711-1719. doi:10.1248/bpb.12-00297.
- Chang H, Sheu M, Yang C, et al. Analgesic Effects and the Mechanisms of Anti-Inflammation of Hispolon in Mice. 2011;2011. doi:10.1093/ecam/nep027.
- Chang WP, Wu JJS, Shyu BC. Thalamic Modulation of Cingulate Seizure Activity Via the Regulation of Gap Junctions in Mice Thalamocingulate Slice. PLoS One. 2013;8(5). doi:10.1371/journal.pone.0062952.
- Friedman M. Mushroom Polysaccharides : Chemistry and. 2016. doi:10.3390/foods5040080.
- Gao L, Sun Y, Si J, et al. Cryptoporus volvatus extract inhibits influenza virus replication in vitro and in vivo. PLoS One. 2014;9(12). doi:10.1371/journal.pone.0113604.
- Geng P, Siu KC, Wang Z, Wu JY. Antifatigue Functions and Mechanisms of Edible and Medicinal Mushrooms. Biomed Res Int. 2017;2017. doi:10.1155/2017/9648496.
- Grienke U, Kaserer T, Pfluger F, et al. Accessing biological actions of Ganoderma secondary metabolites by in silico profiling. Phytochemistry. 2015;114:114-124. doi:10.1016/j.phytochem.2014.10.010.
- Griffin, David H. (State University Of New York, U. Usa. Fungal Physiology. John Wiley And Sons, 1996.
- Guggenheim AG, Wright KM, Zwickey HL. Immune Modulation From Five Major Mushrooms: Application to Integrative Oncology. Integr Med. 2014;13(1):32-44
- Hong D, Park MINJU, Jang EUNH, Jung BOM, Kim NAMJ, Kim JHO. Hispolon as an inhibitor of TGF ‑ β ‑ induced epithelial ‑ mesenchymal transition in human epithelial cancer cells by co ‑ regulation of TGF ‑ β ‑ Snail / Twist axis. 2017:4866-4872. doi:10.3892/ol.2017.6789.
- Hsieh M, Chien S, Chou Y, Chen C, Chen J, Chen M. Phytomedicine Hispolon from Phellinus linteus possesses mediate caspases activation and induces human nasopharyngeal carcinomas cells apoptosis. Eur J Integr Med. 2014;21(12):1746-1752. doi:10.1016/j.phymed.2014.07.013.
- Huang G, Hsieh W, Chang H, Huang S, Lin Y, Kuo Y. r -Glucosidase and Aldose Reductase Inhibitory Activities from the Fruiting Body of Phellinus merrillii. 2011:5702-5706. doi:10.1021/jf2003943.
- Jahromi SS, Wentlandt K, Piran S, Carlen PL. Anticonvulsant actions of gap junctional blockers in an in vitro seizure model. J Neurophysiol. 2002;88(4):1893. doi:10.1152/jn.00801.2001.
- Jin X, Ruiz Beguerie J, Sze DM-Y, Chan GCF. Ganoderma lucidum (Reishi mushroom) for cancer treatment. Cochrane database Syst Rev. 2016;4:CD007731. doi:10.1002/14651858.CD007731.pub3.
- Manjarrez-Marmolejo J F-PJ. Gap Junction Blockers: An Overview of their Effects on Induced Seizures in Animal Models. Curr Neuropharmacol. 2016;14(7):759-771. doi:10.2174/1570159X14666160603115942.
- Melappa G, Roshan A, Nithi C, Mohummed TS, – C, Poojari CC. Phytochemical analysis and in vitro antioxidant, antimicrobial, anti-inflammatory and cytotoxicity activities of wood rotting fungi, Trametes ochracea. Pharmacogn J. 2015;7(2):136-146. doi:10.5530/pj.2015.2.8.
- Morath SU, Hung R, Bennett JW. Fungal volatile organic compounds: A review with emphasis on their biotechnological potential. Fungal Biol Rev. 2012;26(2-3):73-83. doi:10.1016/j.fbr.2012.07.001.
- Plummer NT. Part 2: Treatments for Chronic Gastrointestinal Disease and Gut Dysbiosis. 2015;14(I):25-33.
- Ren G, Liu XY, Zhu HK, Yang SZ, Fu CX. Evaluation of cytotoxic activities of some medicinal polypore fungi from China. Fitoterapia. 2006;77(5):408-410. doi:10.1016/j.fitote.2006.05.004.
- Rep NP. Traversing the fungal terpenome. 2015;31(10):1449-1473. doi:10.1039/c4np00075g.Traversing.
- Ríos JL. Effects of triterpenes on the immune system. J Ethnopharmacol. 2010;128(1):1-14 doi:10.1016/j.jep.2009.12.045.
- Rösecke J, Pietsch M, König WA. Volatile constituents of wood-rotting basidiomycetes. Phytochemistry. 2000;54(8):747-750. doi:10.1016/S0031-9422(00)00138-2.
- Schmidt-dannert C. Biosynthesis of Terpenoid Natural Products in Fungi. 2015;(November 2014):19-61. doi:10.1007/10.
- SCHRADER, JENS. BIOTECHNOLOGY OF ISOPRENOIDS. SPRINGER INTERNATIONAL PU, 2016.
- Shamtsyan M, Antontceva E, Panchenko A, Petrischev N. HYPERLIPIDEMIC AND HYPOCHOLESTEROLIC ACTION OF SUBMERGE CULTURED MUSHROOMS.
- Sumner JL. The fatty acid composition of basidiomycetes. 2012;8643(1973). doi:10.1080/0028825X.1973.10430293.
- Taborda CP, Janeiro R De. GLYCOCONJUGATES AND POLYSACCHARIDES OF FUNGAL CELL WALL AND ACTIVATION. 2008:195-208.
- Teng F, Bito T, Takenaka S, Yabuta Y, Watanabe F. Vitamin B 12 [ c ‑ lactone], a Biologically Inactive Corrinoid Compound, Occurs in Cultured and Dried Lion ’ s Mane Mushroom ( Hericium erinaceus ) Fruiting Bodies. 2014. doi:10.1021/jf404463v.
- Teplyakova T V., Psurtseva N V., Kosogova TA, Mazurkova NA, Khanin VA, Vlasenko VA. Antiviral Activity of Polyporoid Mushrooms (Higher Basidiomycetes) from Altai Mountains (Russia). Int J Med Mushrooms. 2012;14(1):37-45. doi:10.1615/IntJMedMushr.v14.i1.40.
- Velíšek J, Cejpek K. Pigments of Higher Fungi : A Review. 2011;29(2):87-102.
- Wu Q, Kang Y, Zhang H, Wang H, Liu Y, Wang J. Biochemical and Biophysical Research Communications The anticancer effects of hispolon on lung cancer cells. Biochem Biophys Res Commun. 2014;453(3):385-391. doi:10.1016/j.bbrc.2014.09.098.
- Younis AM, Wu F-S, El Shikh HH. Antimicrobial Activity of Extracts of the Oyster Culinary Medicinal Mushroom Pleurotus ostreatus (Higher Basidiomycetes) and Identification of a New Antimicrobial Compound. Int J Med Mushrooms. 2015;17(6):579-590. doi:10.1615/IntJMedMushrooms.v17.i6.80.
- Yu Z, Liu B, Mukherjee P, Newburg DS. Trametes versicolor Extract Modifies Human Fecal Microbiota Composition In vitro. 2013:107-112. doi:10.1007/s11130-013-0342-4.
- Zhu Q, Bang TH, Ohnuki K, Sawai T, Sawai K, Shimizu K. Inhibition of neuraminidase by Ganoderma triterpenoids and implications for neuraminidase inhibitor design. Sci Rep. 2015;5(AUGUST):13194. doi:10.1038/srep13194.